AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Synergy pharmaceuticals8/23/2023 
These limits are being/have been addressed:.Very limited preferred commercial insurance coverage.With only a single indication on offer (CIC).Only a single drug (a byproduct of not being partnered).While it can be stated that the rollout has been sluggish, considering the small sales force (as mentioned earlier) as well as a litany of other barriers to growth listed below: After many weeks of data since launch, nothing indicates that the compound is not being received well by its end users, the patient. The road for its mid and long-term potential remains as good as it ever was, albeit on a more elongated path. Although we view product as approvable (assuming success in T3MPO-2, reading out next half) the results lower our estimates and partnership interest for the drug, particularly in light of the Trulance launch.”Īll things considered, the purpose of this revisit is more so to isolate the performance of Trulance from all the macro noise. “… looked the data over and concluded that the drug looked approvable, but not a top performer, landing somewhere in between Linzess and Trulance (generally not as efficacious as Linzess or as well-tolerated as Trulance). 2.6% for Trulance and 4.5% for Linzess), the jury is out on whether doctors, once familiarized with Trulance (not to mention already with Linzess) will be open to prescribing an (inferior) alternative.Īlthough Tenapanor represents further unneeded competition, Trulance remains the better option as per Wedbush’s David Nierengarten who stated in Q2 2017: But with a diarrhea rate of 14.6% in trial 1 and 16% for trial 2 (causing a 5.9% discontinuation rate vs. (NASDAQ: ARDX), which had a second set of successful Phase 3 results from its IBS-C candidate Tenapanor announced back in October. The catch here being that while SCMP has a very promising pipeline (outlined in this article ), its CIC/IBS-C/OIC lead compound Amitiza has only a few years until patent expiration another benchmark upon which to measure Synergy’s desirability and perceived value from a third party.įinally, Ardelyx Inc. The other news was the acquisition of Synergy competitor Sucampo Pharmaceuticals (NASDAQ: SCMP) for $1.2B by UK-based Mallinckrodt Plc (NYSE: MNK). The coming weeks will indicate whether the change was simply a shuffle to appease justifiably frustrated investors or the mark of something much more fruitful. 
This news yielded some investor confidence as the stock price cleared and has stayed over the $2 mark since. Jacob stays on as Executive Chairman of the Board (formerly Chairman) in this rearrangement. Since then, there have been some developments that could influence the coming months.įor one, the CEO and co-founder Gary Jacob was announced on Dec 23rd to be suddenly stepping down in favor of being replaced by Chief Commercial Officer Troy Hamilton. SGYP struck its 52-week low (and multiyear low of $1.68) just last month along with one of the biggest short positions in the market upon news of yet another dilution. Unfortunately, up until this point in time, investor returns in SGYP have not exactly been ideal to say in the very least. We have demonstrated the potential of having an investment in Synergy in several other articles, here and here, by thoroughly describing what the overall environment in the Gastrointestinal (GI) market is along with Ironwood’s (NASDAQ: IRWD) lead drug Linzess. Although having only been approved for the first of these two indications (with IBS-C PDUFA set for Jan 24th 2018) and a marginal 250-person sales force, Trulance has managed to build up a solid base of prescriptions over the last nine months.  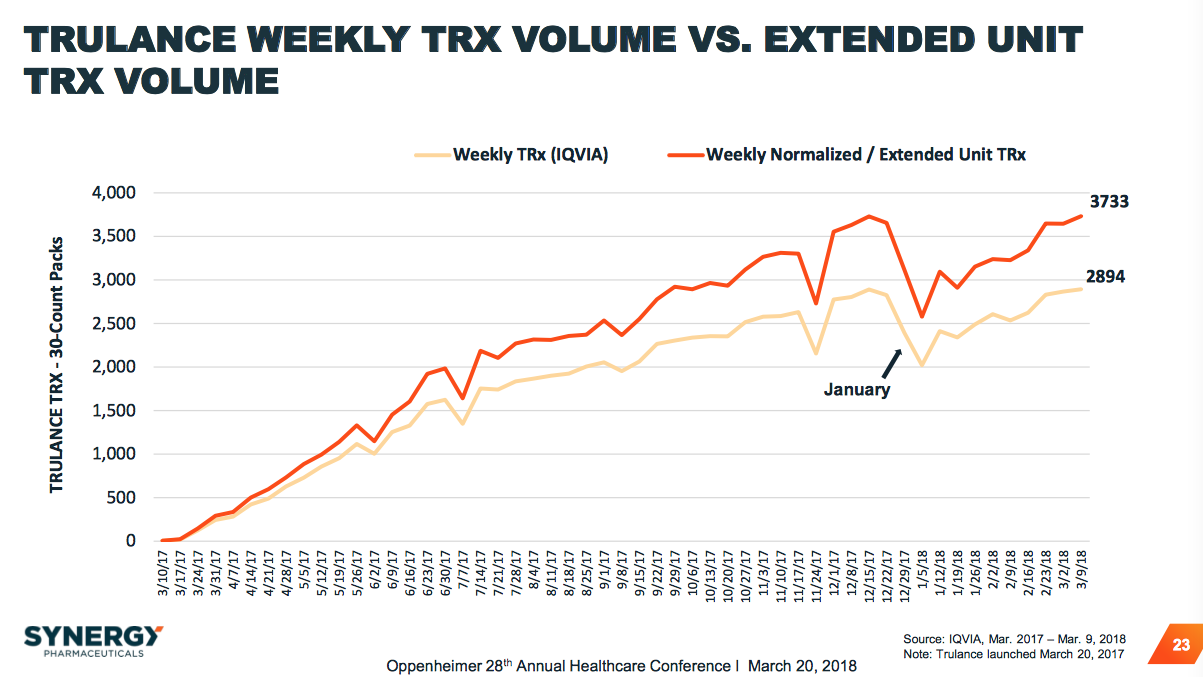
As of this coming March, Synergy Pharmaceuticals (NASDAQ: NASDAQ: SGYP-OLD) will mark the one-year anniversary of the launch of its first FDA approved product, Trulance, for the treatment of Chronic Idiopathic Constipation (CIC) and Irritable Bowel Syndrome – Constipation (IBS-C).
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
